 Market analysis and information related to key drivers, restraints and opportunities are provided. 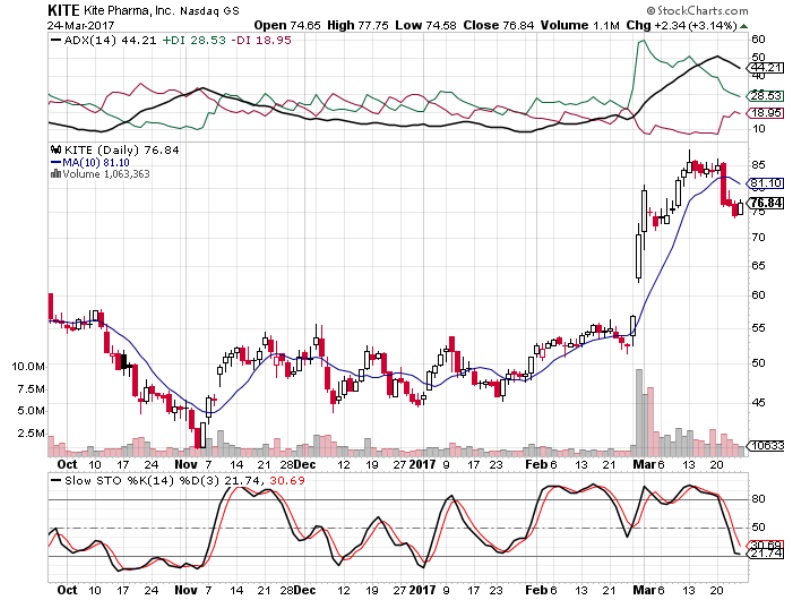
In-depth analysis of this sector helps identify current market opportunities. The report provides quantitative analysis of market segments, current trends, strategies and potential of car t-cell therapy market research to identify potential car t-cell therapy market opportunities in genetics. Major market players covered in the report, such as -Ĭelyad, Gilead Sciences, Inc. Globally, the CAR T-cell therapy market share is witnessing significant growth, owing to increase in technological advancements, increase in prevalence of cancer, and favorable reimbursement policies in some countries, which are expected to create new opportunities for CAR T-Cell therapy market growth during the forecast period. The global car t-cell therapy market size was valued at $1.7 billion in 2021, and is projected to reach $6.1 billion by 2031, growing at a CAGR of 13.5% from 2022 to 2031. This innovative immunotherapy has demonstrated remarkable success in treating certain types of leukemia and lymphoma, achieving deep and durable responses in patients who have exhausted other treatment options. Chimeric Antigen Receptor T-Cell (CAR T-cell) therapy involves genetically engineering a patient's own T-cells to target and destroy cancer cells. PORTLAND, OREGON, UNITED STATES, September 5, 2023/EINPresswire/ - The CAR T-Cell therapy market represents a groundbreaking frontier in the field of cancer treatment, offering a highly promising approach to combat variforms of malignancies. CAR T-cell therapy is a method of training the immune system to recognize cancercells. A research deal with Arcus Biosciences targeting another controversial cancer pathway called TIGIT hasn’t made much progress, and a collaboration with European biotech Galapagos has run into trouble. Gilead’s efforts to develop an oncology business began more than a decade ago when it bought out Calistoga Pharmaceuticals for $375 million, which gave it a blood cancer drug called Zydelig that has largely disappeared from use. Combined, its three marketed cancer drugs are on track to exceed $2 billion in sales this year, but they’re overshadowed by Gilead’s antivirals business, which between HIV, hepatitis and COVID drugs racked up more than $11 billion in sales in the first six months of 2023. The hold is also a blow for Gilead’s strategy to build an oncology business beyond the CAR-T drugs it acquired with Kite Pharma and breast cancer drug Trodelvy, which came via the $21 billion buyout of Immunomedics. Trials in both AML and myelodysplastic syndrome were put on clinical hold in early 2022, but were allowed to proceed a few months later. Like CD47 projects that were canceled, magrolimab has had a bumpy road in testing so far. Big pharma paid handsomely for rights to CD47 drugs, including Gilead’s takeout of Forty Seven, Pfizer’s $2.3 billion acquisition of Trillium AbbVie’s licensing deal for I-Mab’s CD47 and Boehringer Ingelheim’s deal to license a CD47 from OSE Immunotherapeutics. One such pathway was CD47, a cell protein that sends away immune cells called macrophages, the so-called “don’t-eat-me” signal. 
Dive Insight:Īfter the success of cancer immunotherapies like Keytruda and Opdivo, drugmakers went on a hunt to find new ways to defeat cancer by turning the immune system onto tumor cells. Earlier this month, a small drug developer called ALX Oncology terminated trials of its CD47 drug in AML and myelodysplastic syndrome, the latter a condition in which magrolimab failed a study in July. The FDA action is another setback for magrolimab’s drug class, called CD47 inhibitors, which has seen Bristol Myers Squibb and Surface Oncology cancel projects. 
Patients already taking magrolimab as part of ongoing AML trials can continue to receive it, and the hold doesn’t affect any trials in solid tumors. 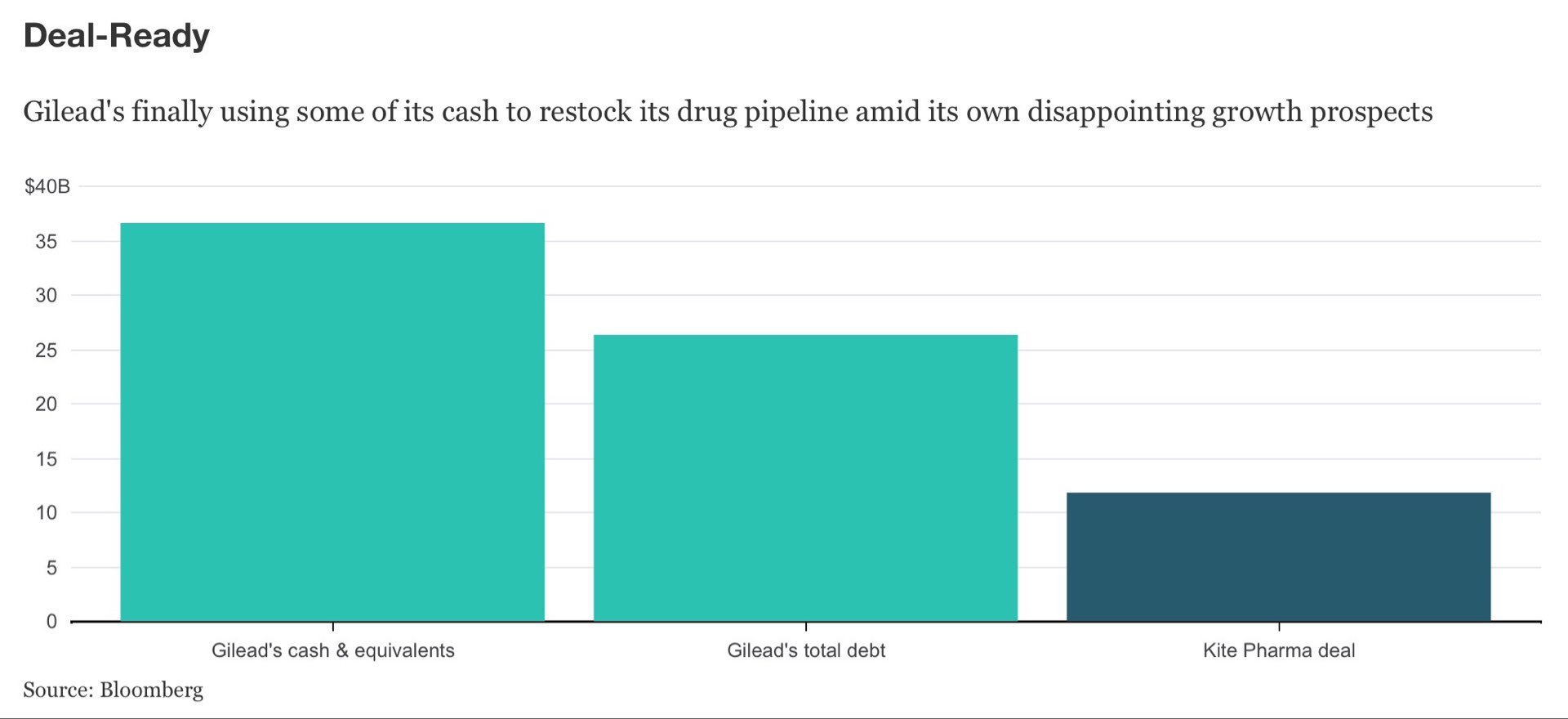
Without detailing the reasons for the partial clinical hold, Gilead said it is discussing next steps with the FDA and other regulators to resolve the hold. The Food and Drug Administration has told Gilead Sciences to suspend patient enrollment in clinical trials of magrolimab in acute myeloid leukemia, the company said Monday, in another setback for an experimental drug the company acquired via a $5 billion buyout three years ago. 21, 2023Ī sign is posted in front of the Gilead Sciences headquarters on April 29, 2020, in Foster City, California. The clinical hold order comes one month after negative results from a study of the CD47 drug, called magrolimab, in myelodysplastic syndrome. FDA partially halts leukemia studies of Gilead cancer drug 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
